Case Study
The Power of Post-Trial Data: Pharma Companies Improve Drug Safety, Efficacy, and Cost Savings by 10% with Comprehensive Analysis using Decision Tree Regression
Platforms – Data Analytics
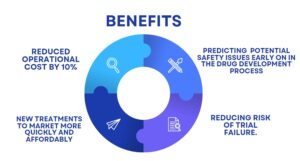
 linical trials cost anywhere from $100,000 to $2 million to over $30 million, including four stages namely Phase I – Trials test a drug’s safety on healthy volunteers. Phase II and III – Trials test the drug’s efficacy on patients. Phase IV – Trials or pharmacovigilance are conducted once the drug is marketed to monitor for its safety in larger populations. This paper focused on leveraging the power of AI, for achieving greater efficiency and accuracy, ultimately bringing new therapies to market more quickly and affordably along with operational cost reduction up to 10% and these techniques can be applied on every phase of trials.
linical trials cost anywhere from $100,000 to $2 million to over $30 million, including four stages namely Phase I – Trials test a drug’s safety on healthy volunteers. Phase II and III – Trials test the drug’s efficacy on patients. Phase IV – Trials or pharmacovigilance are conducted once the drug is marketed to monitor for its safety in larger populations. This paper focused on leveraging the power of AI, for achieving greater efficiency and accuracy, ultimately bringing new therapies to market more quickly and affordably along with operational cost reduction up to 10% and these techniques can be applied on every phase of trials.
Problem Statement:
Developing a model that can accurately predict potential safety issues early on in the drug development process by analyzing the kpis and patient data from every phase ,reducing clinical trial costs by up to 10%.
Expected benefits:-
Data Required:
This table provides an overview of different data points related to demographics, health measurements, and key indicators that can be useful for model building and evaluating health-related metrics.
| Sr. No | Data Point | Source |
| 1 | Age, Gender, and Other Demographic Data | ERP sales module |
| 2 | Gender | ERP sales module |
| 3 | Height | Periodical Tests |
| 4 | Weight | Periodical Tests |
| 5 | Body Fat % | Periodical Tests |
| 6 | Blood Pressure | Periodical Tests |
| 7 | Cholesterol | Periodical Tests |
| 8 | Glucose Level | Periodical Tests |
| 9 | BMI | KPI |
| 10 | Cardio Score | KPI |
KPI Identified:
Cardio Score = (Excess Body Fat*0.25) + (Excess Cholesterol*0.25) + (BMI Increase or Decrease*0.25) + (BP Range*0.25)
The above score was formulated giving equal importance to all the contributors of cardiac diseases. Lesser the cardio score, healthier the person’s heart is. The formula can be modified according to the user’s wants and needs.
Steps towards solution
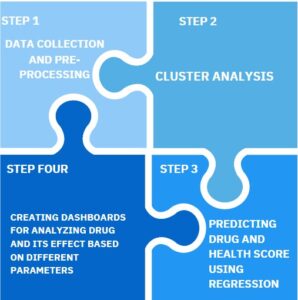
To initiate with we collected data using different demographics point along with kpi to perform exploratory data analysis clubbed with null and outlier treatment Post that we performed custer analysis to understand similarities and differences between grouped formed we used techniques such as hierarchical clustering, db scan along with feature engineering and optimization clustering technique by parameter tuning.We used various regression models to predict the drug score and medical score namely random forest , svc, gradient boosting , etc and we concluded that Decision tree algorithm performed best with accuracy of 88%. Lastly, we developed a dashboard using GroundZero Unified Data platform, focusing on effectiveness of drugs over every phase of the trial.
Dashboard for CRDO:
The CRDO dashboard gives a holistic view about the consumers along with the drug efficiency clustered into 3 categories: healthy , normal, risky.
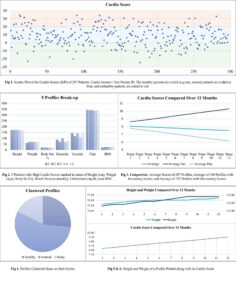
Inference:
A new score was formulated and plotted in a scatter graph which clusters the test patients into three categories. The break-up is shown in the pie-chart.
Patients with high cardio scores were extracted and examined to find the area of the problem. High cholesterol was found to be the root cause.
Average of the total scores, scores that are increasing, and scores that are decreasing was compared. This gave an inference about how well the drug is responding positively and negatively.
If one of the patients is to be extracted his or her cardio score can be compared with their physical wellness.
Short-term impacts:
- Improved accuracy and efficiency can help pharmaceutical companies identify safety issues early in the drug development process, reducing the need for extensive and costly research and development (R&D). The immediate impact includes cost savings (from 5 Million USD to at least 4 Million USD) in the R&D phase and a more streamlined drug development process.
- By optimizing the use of resources in clinical trials, pharmaceutical companies can reduce waste and improve overall sustainability. This can result in immediate improvement in operational efficiency.
Long-term impacts:
- Better insights into patient demographics and needs can enable pharmaceutical companies to develop drugs that are more tailored to individual patients. This can lead to increased sales by 10% in the long term as personalized medications have the potential to address specific patient needs more effectively.
- The ability to reduce average clinical trial costs from 5 million to 4.5 million can have long-term financial benefits for pharmaceutical companies. It can lead to cost savings across multiple drug development projects, contributing to improved profitability and sustainability over time.
Conclusion:
To sum up, the use of machine learning-powered dashboards can be a game-changer for the pharmaceutical industry, offering benefits such as cost reduction, improved safety and efficacy of drugs, and faster time-to-market. These techniques can be applied at every phase of clinical trials, making the process more efficient and effective. This solution was developed on the GroundZero Unified Data platform.
